Structure and characterization of micelles and liposomes
The selective release of a drug substance at the intended location is known as drug targeting. In this process, active substances are bound to a carrier and transported to the intended place of effect. The selectivity is ensured by an adapted surface chemistry and the size of the carrier [1].
The advantages of drug targeting over conventional methods of application are faster and better bioavailability, lower dosage, fewer side effects and longer shelf life due to the encapsulation of the active ingredients. On the other hand, there is the difficulty of finding suitable carriers and incorporating the active ingredients into the carrier.
Nanoparticulate systems are often used as carriers, for example nanoparticles, liposomes and micelles. Liposomes and especially micelles are very suitable for drug targeting of cannabis oil (cannabidiol (CBD)), for example. The absorption of CBD can be greatly increased by nano-encapsulation.
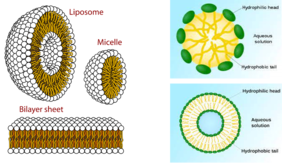
Liposomes are hollow spheres that enclose an aqueous or polar phase through a double lipid layer. In contrast, micelles have only one lipid layer and the inner phase is lipophilic (Figure 1).
Liposomes can also store lipophilic substances in the bilayer. The bilayer usually consists of phospholipids (e.g. lecithin), where the functional groups determine the interaction with the body's own cells and can be specifically adapted by suitable choice of ligands on the phosphate group. Liposomes and micelles are usually produced by energy application, for example by ultrasound treatment [2], which produces very homogeneous dispersions.
Besides the surface chemistry, the size of the carrier systems is of decisive importance for the bioavailability as it enables or limits the migration of the liposomes through the physiological barriers.
A direct ingestion of CBD only leads to a resorption of 10-20% of the active substance. This value can be increased up to 90% by nanocapsulation [3,4]. The particle sizes of liposomes are in the range of 50 - 1000 nm; for optimal bioavailability in oral administration of CBD, the sizes should preferably be between 5 and 50 nm. Micelles usually have sizes below 50 nm. Therefore, micelles are commonly used for CBD encapsulation.
The energy input to achieve the desired particle size should be as low as possible to minimise degradation of the active ingredient during processing.
A useful method to ensure this is the inline measurement of the particle size using DLS, which enables precise monitoring and control of the manufacturing process.
Results of the inline particle size measurement of micelles
The production of the micelles with lecithin was carried out by ultrasonic treatment with a Hielscher ultrasonic device (UP200St and sonotrode S26d14 (100% = 90 µm amplitude)). The particle sizes were determined with the NANO-flex II incl. IPAS extension (configuration Fig. 2) in the flow-through at different energy impacts.
Based on the special measuring head design, the IPAS makes it possible to carry out measurements in flowing liquids. Due to the short measuring time (approx. 30 s), a direct determination of the particle size during the manufacturing process is guaranteed. Through various interfaces of the IPAS, the result of the measurement can be used to control the process. This reduces process costs and prevents a long process duration, which can lead to a degradation of the active ingredients.
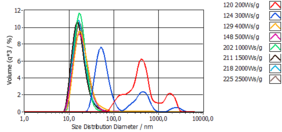
Figure 3 shows the volume distribution of the micelles as a function of the energy input. Up to 500 Ws/g a significant decrease in particle size can be observed, higher energy inputs have no influence on the particle size distribution.
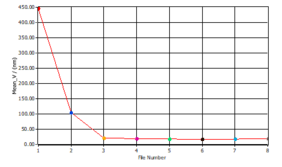
This is apparent when the mean particle size is plotted against the energy input.
Energy inputs above 500 Ws/g do not change the particle size distribution of the micelles further.
The measuring probe of the NANO-flex II can be integrated into the process by the IPAS extension. This eliminates the need for time-consuming sampling. In addition, no cuvettes are necessary. There is no time delay in determining the particle size.
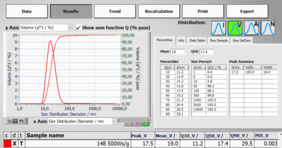
The final result is directly available (Figure 4).
The combination of NANO-flex® II and IPAS in the production of micelles/liposomes and nanoemulsions offers a number of advantages over conventional DLS instruments:
- Due to the 180° heterodyne measurement method, even high concentrations can be measured with high resolution without multiple scattering
- Direct acquisition of measured values without time delay
- The flexible probe makes cuvettes obsolete, the measurement takes place directly in the process
- The measured data can be used for process control via interface
- Zero adjustment (Setzero) can be carried out directly in the reaction vessel
- Simple, uncomplicated software and convenient data transfer simplify operation
Of course, the NANO-flex II can also be used without the IPAS extension for measurements in the laboratory. The flexible probe eliminates the need for cuvettes in this case as well.
Download
Sources
[1] https://en.wikipedia.org/wiki/Targeted_drug_delivery
[2] https://www.hielscher.com/ultrasonic-cannabis-oil-emulsion.htm
[3] https://dremekblair.com/wp-content/uploads/2020/05/Liposomal-Cannabidiol-Delivery-AJEM-2.pdf